Types of Microscopes for Cell Observation
The optical microscope is a useful tool for observing cell culture. However, successful application of microscope observation for culture evaluation is often limited by the skill of the operator and/or the lower reproducibility of visual evaluations. Automatic imaging and analysis for cell culture evaluation helps address these issues, and is seeing more and more practical use. This section introduces microscopes and imaging devices commonly used for cell culture observation work. Key basic microscope components and different types of microscope system configurations/structures will be explored.
Addressing the Challenges of Cell Evaluation using Image Analysis Technology
The optical microscope is a useful tool for observing cell culture and confirming that they are proceeding properly.
An operator visually observes cultured cells using an optical microscope in order to evaluate their condition and determine whether to proceed to the next step in the culture process. However, since this judgment is based on visual evaluation, inconsistencies in judgment will become manifest with multiple operators. Even skilled operators will exhibit a certain amount of variability in their judgment. For this reason, accurate evaluation of cells is a skill that requires years of experience to develop.
Applications for cultured cells are expected to continue to grow in areas such as drug discovery screening, safety evaluation, regenerative medicine, and basic research. Consequently, there is a need for an objective and stable evaluation methodology to replace visual evaluation, which continues to be pervasive.
Thanks to innovations in computer technology, image acquisition and analysis techniques continue to advance dramatically. Cell image data provides a permanent record that can always be referred to. Image analysis may be applied towards quantitative and unbiased evaluation of cell state, morphology, and behavior over time.
This technological evolution has made visual observation obsolete for so much of today’s microscope-based work. Automatic image acquisition by a cell observation system combined with image analysis for culture evaluation has become a reality. By using such unbiased and reproducible evaluation methods, not only can cells be cultured with consistent quality, but evaluation is no longer dependent on the skill and experience of human operators. This also enables more efficient training of operators.
Configuration of an Image Analysis-Ready Microscope
Captured cell images are used as the raw data for quantitative evaluation via image analysis. For that reason, it should go without saying that the quality of the captured images is of high importance. There are many and complex factors affecting image quality that the operator must correctly understand and account for before acquiring images.
Microscopes are built in a modular fashion, with each part having a defined purpose and combining to form a complete system. Optimizing image quality requires both understanding the complete configuration of the system and the appropriate setting for each component. In this section, we will briefly explain the key components of an optical microscope. Please note that the part of the optical path providing illumination may be referred to as the “illumination optics” and the part collecting the light from the specimen the “detection optics.”
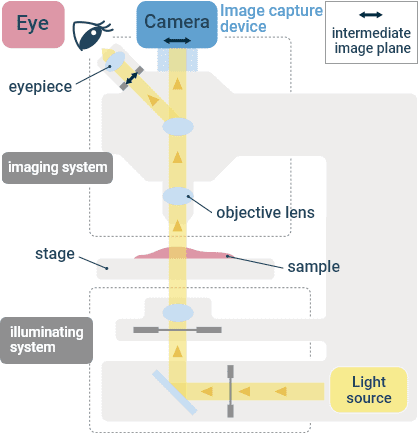
Basic Microscope Configuration
Light Source
Halogen lamps and LEDs are most often used for transmitted light observation, mercury lamps and LEDs for fluorescence, and lasers for confocal microscopy. Since cells are vulnerable to light, care must be taken with the illumination intensity to limit phototoxicity.
Diascopic Illumination Optical System
The transmitted (diascopic) light path usually contains a color adjustment filter, neutral density (ND) filter(s) for brightness adjustment, aperture stop, field stop, condenser, and other components. These components must be aligned and adjusted to provide optimal illumination light flux. Specific contrasting techniques may require additional components, such as prisms for differential interference contrast and phase rings for phase contrast observation.
Objective Lens
Magnification, numerical aperture, immersion medium, chromatic aberration, flatness of field, and other considerations determine the performance of an objective lens. A large variety of lenses for different applications are available from numerous manufacturers. Numerical aperture and refractive index are what determine brightness and resolution, not the magnification. If high resolution is required, use an objective lens designed for immersion in a high refractive index liquid medium, such as water or oil. Such immersion objectives are able to provide higher numerical apertures than air objectives. In general, the size of the field of view and the resolution have a reciprocal relationship.
A special lens with an internal phase film is used for phase contrast observation. For differential interference observation, a prism is inserted into the appropriate objective nosepiece position. In general, high-magnification lenses have shorter working distances (which is the distance from the front lens to the closest part of the observation vessel), so an inverted microscope may not be suitable for imaging through the thick bottom plate of standard culture vessels. When observing three-dimensional specimens such as spheroids, the depth of field needs to be considered as well.
Stage
Sample containers such as glass slides, dishes, flasks, and well plates should be properly secured to the microscope stage. For live cell imaging, install an incubator to control the temperature, humidity, and CO2 concentration in order to maintain cell activity.
Image Acquisition Equipment
In addition to visual observation, a digital camera is often installed on the microscope for acquiring images. Scientific cameras for widefield microscopy usually use either a CCD or CMOS sensor. The sensor converts light energy into electrical energy for image readout. Pixel size and sensitivity are important criteria to consider when selecting a camera. There are monochrome cameras and color cameras, with monochrome cameras providing superior sensitivity. Color reproduction is important for color cameras. In a confocal microscope, the signal from each point is recorded sequentially onto a single element detector such as a PMT.
Types of Microscopes for Collecting Imaging Data
Upright Microscopes

Example of how a typical upright microscope appears
This is a type of microscope in which the specimen is illuminated using a condenser lens below the stage, and observed using an objective lens above the stage. In most cases, the observation target is a fixed specimen on a glass slide, but certain biological specimens require an upright microscope since the objective lens must be directly immersed in the specimen medium. Various microscope components are available to support different contrasting techniques, such as transmitted light (brightfield) and fluorescence observation. With fluorescence observation, the excitation illumination is focused in the specimen using the same objective lens that collects the fluorescence emission.
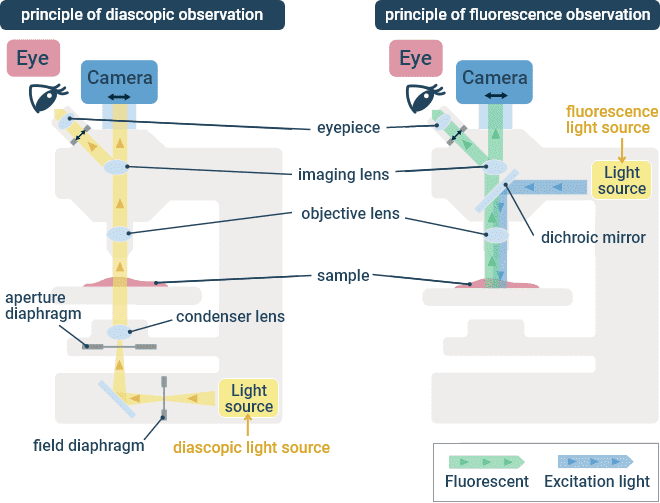
Transmitted Light (Left) and Fluorescence (Right) Optical Paths in an Upright Microscope.
Inverted Microscopes

Example of how a typical inverted microscope appears
The optical arrangement of an inverted microscope is equivalent to that of an upright microscope, except the optical path is flipped such that the specimen is illuminated from above and observed from below. The observation target can also be a fixed specimen on a glass slide, but more often inverted microscopes are used for observing cultured cells in dishes. It is a common tool in basic research, drug discovery screening, safety evaluation, and also the manufacturing process of regenerative medicines. Objective lenses are generally interchangeable on upright and inverted microscopes produced by a common manufacturer. Various microscope components are available to support different contrasting techniques, such as transmitted light (brightfield) and fluorescence. With fluorescence, the excitation illumination is focused in the specimen using the same objective lens that collects the fluorescence emission.
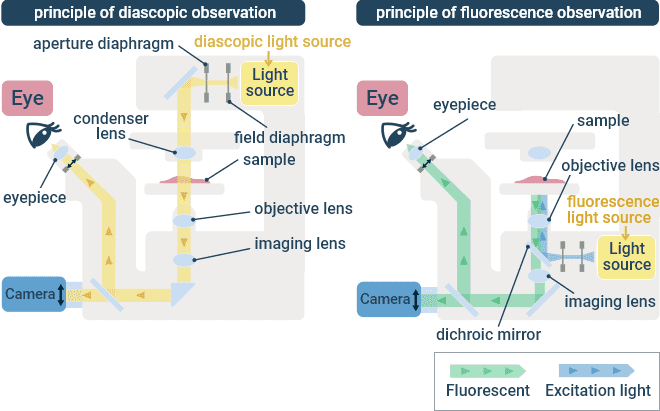
Transmitted Light (Left) and Fluorescence (Right) Optical Paths in an Inverted Microscope.
Stereomicroscopes
Example of how a typical stereomicroscope appears
Stereomicroscopes are oriented such that the specimen is imaged from above, similar to upright microscopes. It has two optical paths, allowing for observation of the specimen from two different angles, which results in a stereoscopic 3D-like view with the illusion of depth. Furthermore, the objective lens can observe specimens across a wider field of view than typical upright or inverted microscopes. It is suitable for performing fine specimen manipulations, such as dissection.
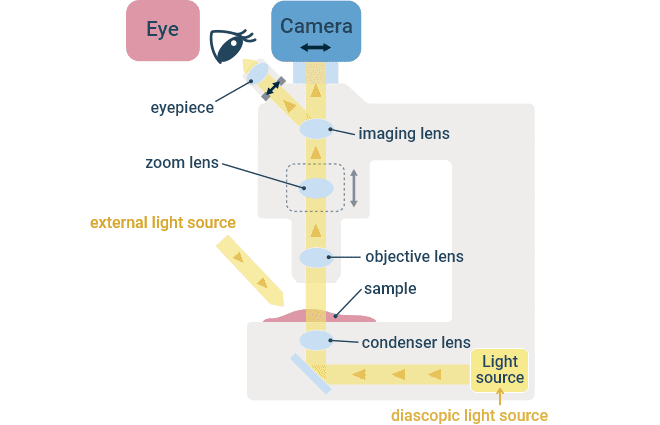
Transmitted Light Path in a Stereomicroscope.
Confocal Microscope
A confocal microscope system mainly consists of a laser light source, confocal scanner, microscope stand, detector system, and computer/software for controlling the various components. It is a large-scale system that scans a laser beam through the sample, detecting the signal at each position sequentially, and using a computer to recombine the information from each position into a complete image. Confocal systems can be configured on both inverted and upright microscope stands, depending on the purpose of the system.

Example of how a typical confocal microscope system appears when configured on an inverted microscope stand.
A confocal microscope uses a laser light source to provide point illumination, and a pinhole is placed prior to the photodetector in a plane conjugate to that of the illuminated point in the specimen. If light is in focus in one plane, it will be in focus in subsequent conjugate planes. Thus the pinhole is a spatial filter that only allows light that was in focus at the objective focal plane to pass, rejecting out-of-focus light and allowing operators to observe discrete optical sections. The laser is focused to a diffraction-limited point in the specimen and raster scanned in the XY directions by the scanner unit. The generated fluorescence emission is captured by the objective lens and descanned by the scanner unit such that the light is always incident on the same location on the detector, regardless of scan position in the specimen. The detector samples the signal at a predefined frequency such that signal can be assigned to single pixels in the resulting image.
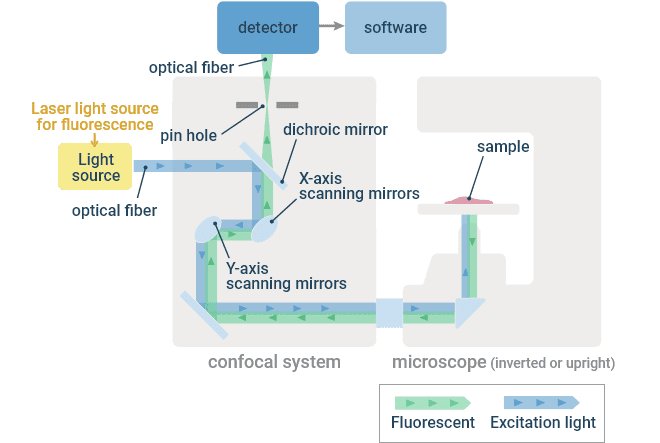
Optical Principle of a Confocal Microscope
The specimen or objective lens can be physically moved in the Z direction to capture images from multiple XY planes, which can then be combined into a 3D representation. This is an effective method for observing the internal 3D structure of cells because it works well even in thick samples.

